Mitochondria Encode Cellular Stress Signals
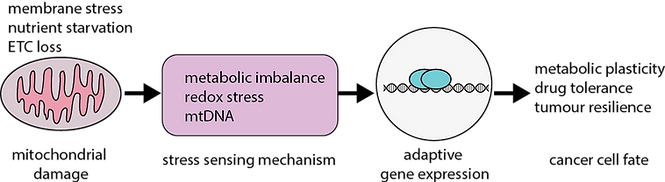
Mitochondria are best known for their bioenergetic roles, but emerging evidence suggests they also function as dynamic stress-sensing and signal transducing organelles. Our research explores how the structure of the mitochondrial inner membranes act as a source of metabolic stress signals that go on to reprogram cell fate. We are particularly interested in how disruptions to cristae organization alter mitochondrial biosynthetic capacity to thereby engage cytoplasmic stress sensing pathways to activate adaptive cellular programs such as the Integrated Stress Response (ISR). These homeostatic pathways allow cells to rewire multiple nodes of the metabolic network to ultimately survive otherwise lethal challenges.
In cancer, these mechanisms may enable tumor cells to enter transient drug-tolerant states, promoting survival during therapy and contributing to relapse. Our vision is to understand how mitochondrial structure and signaling shape cellular resilience—and how these pathways might be targeted to improve cancer treatment.
we are currently looking for new graduate students interested in
cancer cell biology
email: eywc@queensu.ca
We thank our current and past funders


